Pioneer Transcription Factors are critical proteins that initiate gene expression in previously inaccessible chromatin regions. Understanding their mechanisms requires sophisticated biochemical and biophysical techniques. This article delves into the methodologies employed to study these essential regulatory proteins, drawing from established protocols in protein biochemistry, molecular biology, and structural biology. These methods, initially developed for general protein and DNA analysis, are now indispensable for dissecting the intricate interactions of pioneer transcription factors with chromatin.
I. Protein Expression, Mutagenesis, and Purification: Foundational Steps in Pioneer Transcription Factor Research
The study of pioneer transcription factors at a molecular level necessitates the production of pure and functional proteins. Recombinant protein expression in Escherichia coli remains a cornerstone technique, offering a scalable and cost-effective approach.
Xenopus laevis Histone Expression and Purification for Nucleosome Reconstitution
Histones, the fundamental building blocks of nucleosomes, are often required in large quantities for chromatin reconstitution studies. Xenopus laevis histones, expressed in E. coli, provide a robust source for such experiments. The process involves bacterial cell culture, induction of protein expression using IPTG, and subsequent cell lysis. Inclusion bodies, containing overexpressed histones, are isolated and rigorously washed to remove contaminants.
 Histone proteins separated by SDS-PAGE.
Histone proteins separated by SDS-PAGE.
Histone proteins are then extracted from inclusion bodies using denaturing agents like guanidine hydrochloride. For studies requiring histone pairs (H2A-H2B and H3-H4), equimolar amounts are combined and refolded through dialysis, gradually reducing the denaturant concentration. Cation-exchange chromatography further purifies these histone pairs, followed by size-exclusion chromatography to ensure homogeneity and proper folding. These purified histones are essential for in vitro nucleosome assembly, mimicking the natural chromatin environment for pioneer transcription factor interaction studies.
OCT4 Protein Production and Variants: A Case Study in Pioneer Transcription Factor Preparation
OCT4, a quintessential pioneer transcription factor, plays a central role in pluripotency and reprogramming. Its study often requires both full-length and modified versions. Commercially available OCT4, such as the Abcam protein fused with a VP16 transactivation domain and a 11R tag, serves specific purposes, particularly in “assembly 1” cryo-EM studies.
For other applications, including “assembly 2” cryo-EM and various biochemical assays, His-tagged OCT4 expressed in E. coli is preferred. Similar to histone purification, OCT4 is initially purified under denaturing conditions from inclusion bodies using Talon affinity resins. Refolding is crucial for restoring protein function, achieved through a series of dialysis steps with decreasing urea concentrations and the inclusion of L-arginine to prevent aggregation.
Mutagenesis Techniques for Pioneer Transcription Factor Studies
To dissect specific domains or binding motifs within pioneer transcription factors like OCT4, site-directed mutagenesis is indispensable. Inverse PCR strategy allows for targeted mutations within the OCT4 gene. Oligonucleotide primers containing the desired mutations are used in PCR to amplify the entire plasmid. Subsequent enzymatic treatments with T4 PNK, T4 DNA ligase, and Dpn I facilitate plasmid recircularization and removal of the original template DNA. Transformation into competent E. coli cells and sequencing verification ensures the generation of desired OCT4 variants with precise amino acid changes, crucial for structure-function studies.
II. Chromatin Assembly and Characterization: Recreating the Pioneer Transcription Factor Target Environment
Pioneer transcription factors function within the context of chromatin. Therefore, in vitro reconstitution of nucleosomes and chromatin arrays is vital for studying their interactions with DNA and nucleosomes.
Histone Octamer Assembly and Purification
The histone octamer, composed of two copies each of H2A, H2B, H3, and H4, is the core particle of the nucleosome. Assembly of histone octamers in vitro is achieved by mixing purified H2A-H2B dimers with H3-H4 tetramers in high salt conditions. The high salt concentration (2M NaCl) facilitates histone complex formation. Size-exclusion chromatography, using columns like Superdex S200 Increase, separates the assembled octamer from excess dimers, ensuring a pure and stoichiometric octamer preparation for nucleosome assembly.
DNA Fragment Preparation: LIN28B and MATN1 as Model Pioneer Transcription Factor Target Sites
Specific DNA sequences are often used to study pioneer transcription factor targeting. LIN28B genomic DNA, a known target of OCT4, and MATN1 sequences selected for the presence of OCT4 motifs, are commonly employed. These DNA fragments, either synthesized or PCR-amplified from genomic DNA, are crucial for assembling defined nucleosomes. Mutations within OCT4-binding sites in LIN28B DNA, such as those in LIN28B-1M, -2M, and -3M variants, are valuable for assessing the sequence specificity of pioneer factor binding and function.
Nucleosome Assembly Protocols: Dialysis-Based Reconstitution
Nucleosome assembly, the wrapping of DNA around the histone octamer, is typically performed using salt dialysis. Equimolar ratios of histone octamer and DNA fragment are mixed in high salt buffer (2M NaCl). Slowly reducing the salt concentration through stepwise dialysis, using a “double bag” method to control the rate of salt removal, facilitates the gradual and controlled wrapping of DNA around the histone octamer. Native PAGE and SYBR Gold staining are used to verify successful nucleosome assembly, confirming the formation of mononucleosomes.
Modified Nucleosome Assembly: Epigenetic Context for Pioneer Factor Activity
Pioneer transcription factor function is often influenced by epigenetic modifications on histones. To study these effects, modified nucleosomes containing specific histone modifications are essential. H3K27ac and H3K27me3 modifications, associated with active and repressive chromatin states respectively, are particularly relevant. H3K27ac nucleosomes are assembled using commercially modified histone octamers. H3K27me3 nucleosomes require more complex approaches, including site-directed mutagenesis to create H3K27C mutants, followed by in vitro trimethylation using the MLA protocol and subsequent purification. These modified nucleosomes allow for direct investigation of how histone modifications impact pioneer transcription factor binding and activity.
Nucleosome Array Assembly: Modeling Higher-Order Chromatin
For studies exploring the influence of pioneer transcription factors on chromatin compaction and higher-order structures, nucleosome arrays are indispensable. Longer DNA fragments containing repeating nucleosome positioning sequences, such as a 1,022-bp region from the LIN28 genomic site, are used. Using a similar double dialysis method as mononucleosome assembly but with a higher histone octamer to DNA ratio (1:5), nucleosome arrays are reconstituted. These arrays mimic the more complex organization of chromatin in vivo and are crucial for studying the collective effects of pioneer factors on chromatin structure.
III. Biophysical and Biochemical Assays: Dissecting Pioneer Transcription Factor Mechanisms
A suite of biophysical and biochemical assays are employed to probe the interactions of pioneer transcription factors with nucleosomes and chromatin.
Cryo-EM Grid Preparation and Data Collection: Visualizing Pioneer Transcription Factor Complexes
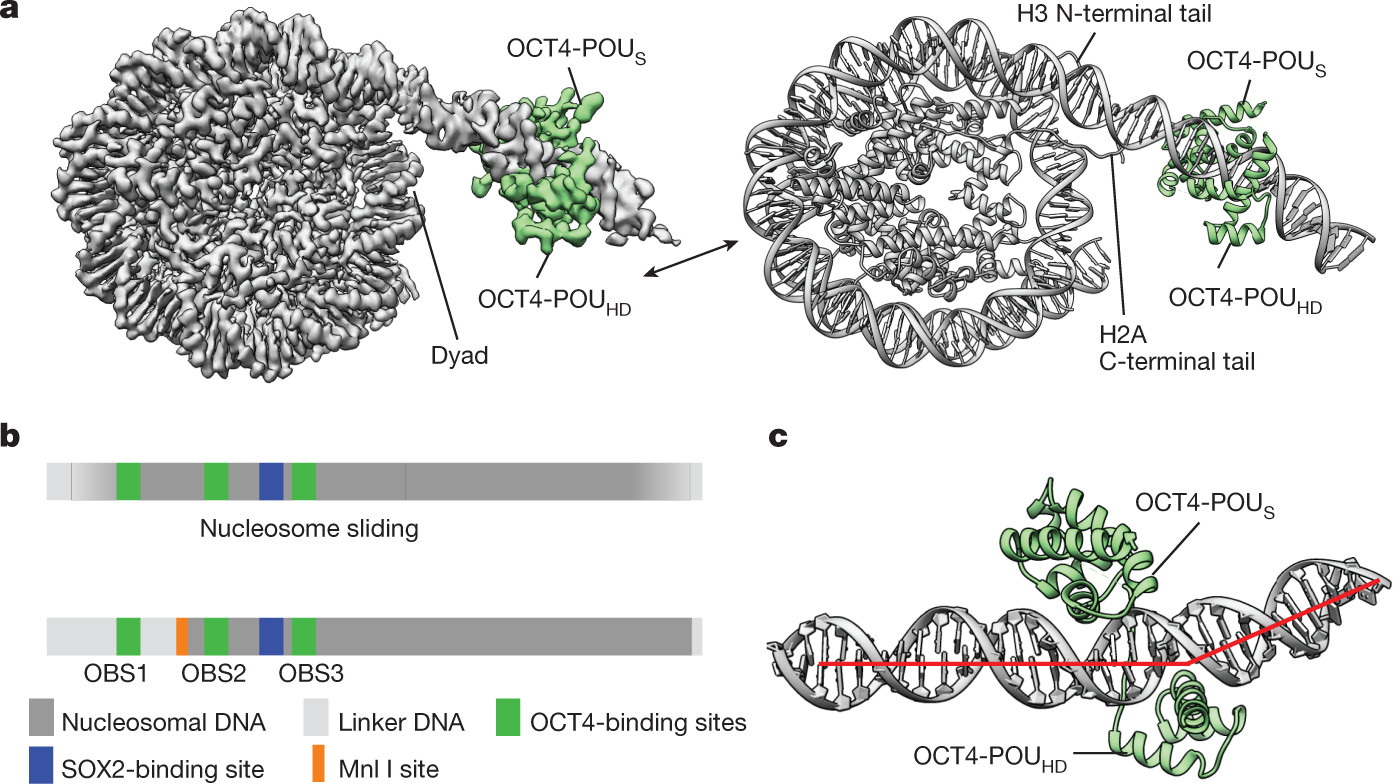
Cryo-electron microscopy (cryo-EM) provides near-atomic resolution structures of macromolecular complexes, including pioneer transcription factors bound to nucleosomes. To prepare samples for cryo-EM, complexes of nucleosomes and pioneer factors like OCT4 and SOX2 are assembled in vitro. For optimal grid preparation, conditions are optimized to minimize aggregation and ensure even particle distribution. Samples are applied to cryo-EM grids, blotted, and plunge-frozen in liquid ethane.
Cryo-EM data collection is performed using high-end electron microscopes, such as FEI Titan Krios, equipped with direct electron detectors like Gatan Summit K3 or Falcon 4. Data are collected at high magnifications and across a range of defocus values. Motion correction and contrast transfer function (CTF) estimation are crucial processing steps before particle picking and 3D reconstruction, ultimately yielding high-resolution structures of pioneer factor-nucleosome complexes.
Restriction Enzyme Digestion Assays: Probing Chromatin Accessibility
Restriction enzyme digestion assays, particularly using enzymes like Mnl I, are used to assess chromatin accessibility in the presence or absence of pioneer transcription factors. Mnl I digests DNA at specific sites, but its access is hindered by nucleosomes. Increased Mnl I digestion in the presence of a pioneer factor indicates increased DNA accessibility, suggesting chromatin remodeling or displacement. Quantitative analysis of digested DNA fragments on polyacrylamide gels provides insights into how pioneer factors alter nucleosome structure and DNA accessibility.
Magnesium Precipitation Assay: Assessing Chromatin Compaction
Magnesium (Mg2+) ions induce chromatin compaction. Magnesium precipitation assays exploit this property to study how pioneer transcription factors influence chromatin folding. Nucleosomes, with or without bound pioneer factors, are incubated with varying concentrations of MgCl2. Centrifugation separates precipitated (compacted) nucleosomes from soluble ones. Quantifying the amount of precipitated nucleosomes reveals the effect of pioneer factors on chromatin compaction, indicating whether they promote or inhibit chromatin folding.
Binding Assays: Quantifying Pioneer Transcription Factor Affinity
Binding assays, such as electrophoretic mobility shift assays (EMSAs), are used to measure the binding affinity of pioneer transcription factors to nucleosomes or DNA. Fluorescently labeled (Cy5-labeled) nucleosomes are incubated with increasing concentrations of pioneer factors. Native PAGE separates bound and unbound nucleosome species. Quantification of band intensities allows for determination of binding constants and stoichiometry, revealing the affinity and cooperativity of pioneer factor-nucleosome interactions.
MNase-seq: Mapping Genome-Wide Pioneer Factor Binding and Chromatin Accessibility
Micrococcal nuclease sequencing (MNase-seq) is a powerful technique to map genome-wide nucleosome positions and chromatin accessibility. When combined with pioneer factor binding studies, MNase-seq reveals how these factors influence chromatin structure at a global scale. Nucleosomes, with or without bound pioneer factors, are digested with MNase, which preferentially cleaves accessible DNA regions. Sequencing the MNase-protected DNA fragments identifies nucleosome positions and accessible regions. Comparing MNase-seq profiles in the presence and absence of pioneer factors reveals their impact on genome-wide chromatin organization and accessibility.
Deacetylation Assays: Investigating Epigenetic Regulation by Pioneer Factors
Deacetylation assays investigate the interplay between pioneer transcription factors and histone acetylation, a key epigenetic modification. H3K27ac nucleosomes, representing an actively transcribed state, are treated with deacetylase complexes like MiDAC. The extent of deacetylation, monitored by western blotting using anti-H3K27ac antibodies, is compared in the presence and absence of pioneer factors. These assays reveal if pioneer factors can modulate histone acetylation levels, potentially influencing gene expression through epigenetic mechanisms.
Negative Stain EM and Image Analysis: Visualizing Chromatin Structure Changes
Negative stain electron microscopy provides a lower-resolution but rapid method to visualize chromatin structure and the impact of pioneer transcription factors. Nucleosome arrays, treated with MgCl2 to induce compaction, are examined with and without pioneer factors. Negative staining with uranyl acetate or uranyl formate enhances contrast, allowing for visualization of chromatin fibers. Image analysis, using software like ImageJ, quantifies particle sizes and compaction levels, revealing the structural consequences of pioneer factor binding on chromatin arrays.
Conclusion: Methodological Arsenal for Unraveling Pioneer Transcription Factor Function
The methodologies outlined here represent a powerful toolkit for studying pioneer transcription factors. From recombinant protein production and nucleosome reconstitution to advanced biophysical and biochemical assays, these techniques provide complementary approaches to dissect the multifaceted mechanisms of pioneer transcription factor action. Integrating structural biology (cryo-EM), chromatin biochemistry (MNase-seq, modification assays), and biophysical measurements (binding assays, compaction assays) is crucial for a comprehensive understanding of how pioneer transcription factors navigate chromatin and initiate gene expression, paving the way for advancements in developmental biology, gene therapy, and regenerative medicine.
References
The references are kept as in the original article, with links provided for easy access.
[48] Luger, K., Rechsteiner, T. J. & Richmond, T. J. Expression and purification of recombinant histones and nucleosome reconstitution. Methods Mol. Biol. 119, 1–16 (1999). ([/articles/s41586-023-06112-6#ref-CR48 “Luger, K., Rechsteiner, T. J. & Richmond, T. J. Expression and purification of recombinant histones and nucleosome reconstitution. Methods Mol. Biol. 119, 1–16 (1999).”)
[49] Ivic, N., Groschup, B., Bilokapic, S. & Halic, M. Simplified method for rapid purification of soluble histones. Croat. Chem. Acta 89, 153–162 (2016). ([/articles/s41586-023-06112-6#ref-CR49 “Ivic, N., Groschup, B., Bilokapic, S. & Halic, M. Simplified method for rapid purification of soluble histones. Croat. Chem. Acta 89, 153–162 (2016).”)
[8] Soufi, A. et al. Pioneer transcription factors target partial DNA motifs on nucleosomes to initiate reprogramming. Cell 161, 555–568 (2015). ([/articles/s41586-023-06112-6#ref-CR8 “Soufi, A. et al. Pioneer transcription factors target partial DNA motifs on nucleosomes to initiate reprogramming. Cell 161, 555–568 (2015).”)
[3] Soufi, A., Donahue, G. & Zaret, K. S. Facilitators and impediments of the pluripotency reprogramming factors’ initial engagement with the genome. Cell 151, 994–1004 (2012). ([/articles/s41586-023-06112-6#ref-CR3 “Soufi, A., Donahue, G. & Zaret, K. S. Facilitators and impediments of the pluripotency reprogramming factors’ initial engagement with the genome. Cell 151, 994–1004 (2012).”)
[50] Bilokapic, S. & Halic, M. Nucleosome and ubiquitin position Set2 to methylate H3K36. Nat. Commun. 10, 3795 (2019). ([/articles/s41586-023-06112-6#ref-CR50 “Bilokapic, S. & Halic, M. Nucleosome and ubiquitin position Set2 to methylate H3K36. Nat. Commun. 10, 3795 (2019).”)
[51] Bilokapic, S., Strauss, M. & Halic, M. Structural rearrangements of the histone octamer translocate DNA. Nat. Commun. 9, 1330 (2018). ([/articles/s41586-023-06112-6#ref-CR51 “Bilokapic, S., Strauss, M. & Halic, M. Structural rearrangements of the histone octamer translocate DNA. Nat. Commun. 9, 1330 (2018).”)
[52] Simon, M. D. et al. The site-specific installation of methyl-lysine analogs into recombinant histones. Cell 128, 1003–1012 (2007). ([/articles/s41586-023-06112-6#ref-CR52 “Simon, M. D. et al. The site-specific installation of methyl-lysine analogs into recombinant histones. Cell 128, 1003–1012 (2007).”)
[53] Carey, M. F., Peterson, C. L. & Smale, S. T. Dignam and Roeder nuclear extract preparation. Cold Spring Harb. Protoc. 2009, pdb.prot5330 (2009). ([/articles/s41586-023-06112-6#ref-CR53 “Carey, M. F., Peterson, C. L. & Smale, S. T. Dignam and Roeder nuclear extract preparation. Cold Spring Harb. Protoc. 2009, pdb.prot5330 (2009).”)
[54] Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012). ([/articles/s41586-023-06112-6#ref-CR54 “Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).”)
[55] Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005). ([/articles/s41586-023-06112-6#ref-CR55 “Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).”)
[56] Grant, T. & Grigorieff, N. Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. eLife 4, e06980 (2015). ([/articles/s41586-023-06112-6#ref-CR56 “Grant, T. & Grigorieff, N. Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. eLife 4, e06980 (2015).”)
[57] Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017). ([/articles/s41586-023-06112-6#ref-CR57 “Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).”)
[58] Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015). ([/articles/s41586-023-06112-6#ref-CR58 “Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).”)
[59] Wagner, T. et al. SPHIRE-crYOLO is a fast and accurate fully automated particle picker for cryo-EM. Commun. Biol. 2, 218 (2019). ([/articles/s41586-023-06112-6#ref-CR59 “Wagner, T. et al. SPHIRE-crYOLO is a fast and accurate fully automated particle picker for cryo-EM. Commun. Biol. 2, 218 (2019).”)
[60] Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife https://doi.org/10.7554/eLife.42166 (2018). ([/articles/s41586-023-06112-6#ref-CR60 “Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife https://doi.org/10.7554/eLife.42166 (2018).”)
[61] Terwilliger, T. C., Ludtke, S. J., Read, R. J., Adams, P. D. & Afonine, P. V. Improvement of cryo-EM maps by density modification. Nat. Methods 17, 923–927 (2020). ([/articles/s41586-023-06112-6#ref-CR61 “Terwilliger, T. C., Ludtke, S. J., Read, R. J., Adams, P. D. & Afonine, P. V. Improvement of cryo-EM maps by density modification. Nat. Methods 17, 923–927 (2020).”)
[62] Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010). ([/articles/s41586-023-06112-6#ref-CR62 “Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).”)
[63] Bilokapic, S., Suskiewicz, M. J., Ahel, I. & Halic, M. Bridging of DNA breaks activates PARP2–HPF1 to modify chromatin. Nature 585, 609–613 (2020). ([/articles/s41586-023-06112-6#ref-CR63 “Bilokapic, S., Suskiewicz, M. J., Ahel, I. & Halic, M. Bridging of DNA breaks activates PARP2–HPF1 to modify chromatin. Nature 585, 609–613 (2020).”)
[64] Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010). ([/articles/s41586-023-06112-6#ref-CR64 “Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).”)
[65] Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004). ([/articles/s41586-023-06112-6#ref-CR65 “Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).”)
